Maine Honey, Maple Syrup Producers Saved from Confusing Labels
August 15, 2018
In recent months, honey and maple syrup producers from the state of Maine have been fighting a US Senate proposal to add a confusing “added sugar” label to all their products, a distinction that could be false in its vague presentation. However, these businesses can rest a bit more easily for now, as an amendment that would prevent the confusing label requirement from going forward has just been approved by the Senate.
This amendment was written by Senator Susan Collins of Maine and cosponsored by her junior senator, Angus King, and they have seemingly protected Maine honey and maple syrup producers from the FDA’s controversial changes. The original requirement was intended to help citizens follow new federal nutrition recommendations and warn them of excess sugar in various products, as it could add to any health concerns, such as diabetes and obesity. However, honey and maple syrup are high in only natural sugars, which is what triggered their adherence to the label requirement, despite neither product containing added sugar like cane sugar or high-fructose corn syrup.
Honey and maple syrup producers were in a tough spot with this requirement, which Mary Anne Kinney of Kinney’s Sugarhouse in Waldo County, Maine described as something that would’ve had “devastating” effects for her family’s business. Maine’s pure honey producers were also worried, as they predicted the labels would’ve worked against their considerable efforts to inform consumers about compromised honey that’s had corn syrup mixed in to increase profit.
Representative Chellie Pingree fought against the FDA’s proposed label requirement by adding language to a House appropriation bill and meeting with FDA Commissioner Scott Gottlieb twice to talk about the issue, with Senator Collins speaking with Gottlieb as well. The public’s response to the FDA’s proposal was so intense the organization had to back off it back in July, saying they would reconsider the proposal after reviewing the feedback they received, which included over 3,000 comments.
According to Collins, “Although the FDA’s rule was well-intentioned, it would have created widespread consumer confusion and negatively affected many family-owned businesses. While I am grateful that FDA has acknowledged the serious concerns expressed in public comments and by members of Congress and has pledged to revise its approach, my amendment will ensure this misguided labeling requirement does not move forward.” Per Collins, her proposal amendment would still offer support for public health and safety by the sugar’s percent daily value remaining on honey and maple syrup labels.
Ready to try some Manuka honey? Click here to get your order started and remember, we offer FREE shipping on all orders of $150 or more!
Photo By aetb

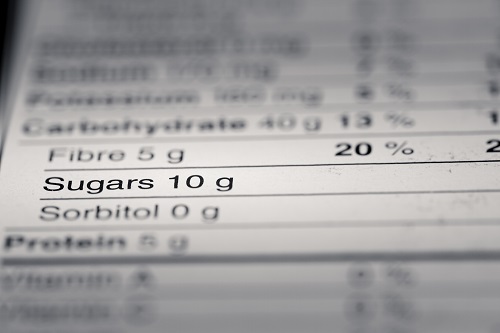
.jpg)




